A recent article in the journal Science announced a discussion of the "Inflammasome" (see the diagram LHS, taken from the Invivogen web site) with the title "Assembling the Wheel of Death". It had to be read! The work was carried out by groups in he USA and China at the Universities of Harvard and Tsinghua. And after reading about this multi-protein complex, I thought it would make an excellent choice for my next Molecule of the Month! So before we look at the molecule itself, just what is the inflammasome and what is its biological role?
Immune defences are possibly amongst the most complex phenomena found in Nature; and from an evolutionary point of view, the sophistication of the immune response is quite different between organisms. In simple terms, the innate immune response is a quick-fire generic mechanism to fend off threats from pathogenic microbes and foreign bodies, such as asbestos fibres. The adaptive immune system, on the other hand, is confined to vertebrates, combats an attack and leaves us able to mount a defence against future attacks of the same kind. The inflammasome is a universal molecular assembly and is part of the innate immune response.
The recent articles in Science provide a structural basis to the triggering of the
assembly of the inflammasome and the image on the right gives you some idea of the overall shape of the complex. The pink "cap" is the NAIP2 protein and the pale blue colour are the NLRC4 proteins. The inflammasome has different components and stoichiometries in different organisms and the organisation shows interspecies variation, that relates to specific differences in the characteristics of the complex in each setting. However, the general features of the inflammasome are shared by all inflammasomes, and I shall concentrate on these here. The first point to note is that the IF (fed up of typing inflammasome already!) is an intracellular complex that promotes the "activation" of proteases and the maturation of cytokines. The immune system is littered with "latent" or "pro" forms of proteins, that require some form of modification, such as the release of an N-terminal peptide, before they can perform their inflammatory-related role. Think of trypsinogen and chymotrypsinogen, pro-enzyme forms of the serine proteases, trypsin and chymotrypsin respectively. The conversion of an inactive form to an active one by limited proteolysis or ligand binding is a common thread in biochemical regulation: the latter is referred to as "allostery" (discussed in an earlier post on RNA Polymerase).
 The image on the left, illustrates the protease cleavage events associated with the activation of Caspase 7, an enzyme (an executioner enzyme, no less) that is intimately associated with programmed cell death (apoptosis). Using cryo-electron microscopy (as an aside, you may have noticed how often this technique is appearing in the structural biology world: it seems to be an unstoppable force and is a major new focus at Sheffield) the authors derive a series of incredibly detailed molecular images. If you are looking for more information, the Wu lab home page contains links to publications that provide a comprehensive background to the molecules and their function. See here, for example.
The image on the left, illustrates the protease cleavage events associated with the activation of Caspase 7, an enzyme (an executioner enzyme, no less) that is intimately associated with programmed cell death (apoptosis). Using cryo-electron microscopy (as an aside, you may have noticed how often this technique is appearing in the structural biology world: it seems to be an unstoppable force and is a major new focus at Sheffield) the authors derive a series of incredibly detailed molecular images. If you are looking for more information, the Wu lab home page contains links to publications that provide a comprehensive background to the molecules and their function. See here, for example.
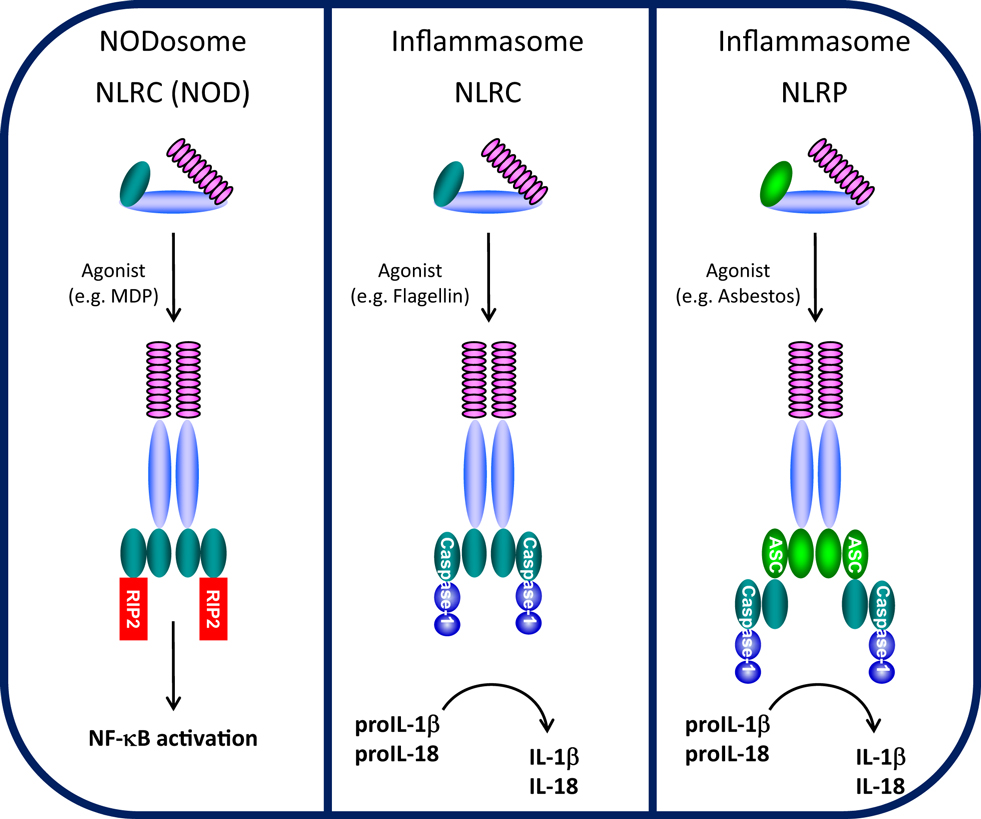 Perhaps the most significant finding of this work (summarised schematically alongside related complexes on the left); notwithstanding the elegant microscopy, is the detail that has revealed how the IF is triggered by a single molecular recognition event from an invading bacterium (the pink blob in the image above), which in turn promotes the assembly of the intact and active IF. In addition, mutations in the NLRC4 protein (the pale blue wheel-shaped assembly attached to the pink cap, above) cause severe autoimmune inflammatory disease. The structure provides insight into how the IF fails to assemble properly in patients carrying these genetic lesions.The use of cryo-electron microscopy, drawing on X-ray crystallography has led to a molecular explanation for the triggering of the inflammatory response and moreover how mutations can throw a spanner in the works of macromolecular assembly, with such a devastating effect.
Perhaps the most significant finding of this work (summarised schematically alongside related complexes on the left); notwithstanding the elegant microscopy, is the detail that has revealed how the IF is triggered by a single molecular recognition event from an invading bacterium (the pink blob in the image above), which in turn promotes the assembly of the intact and active IF. In addition, mutations in the NLRC4 protein (the pale blue wheel-shaped assembly attached to the pink cap, above) cause severe autoimmune inflammatory disease. The structure provides insight into how the IF fails to assemble properly in patients carrying these genetic lesions.The use of cryo-electron microscopy, drawing on X-ray crystallography has led to a molecular explanation for the triggering of the inflammatory response and moreover how mutations can throw a spanner in the works of macromolecular assembly, with such a devastating effect.
Immune defences are possibly amongst the most complex phenomena found in Nature; and from an evolutionary point of view, the sophistication of the immune response is quite different between organisms. In simple terms, the innate immune response is a quick-fire generic mechanism to fend off threats from pathogenic microbes and foreign bodies, such as asbestos fibres. The adaptive immune system, on the other hand, is confined to vertebrates, combats an attack and leaves us able to mount a defence against future attacks of the same kind. The inflammasome is a universal molecular assembly and is part of the innate immune response.
The recent articles in Science provide a structural basis to the triggering of the
assembly of the inflammasome and the image on the right gives you some idea of the overall shape of the complex. The pink "cap" is the NAIP2 protein and the pale blue colour are the NLRC4 proteins. The inflammasome has different components and stoichiometries in different organisms and the organisation shows interspecies variation, that relates to specific differences in the characteristics of the complex in each setting. However, the general features of the inflammasome are shared by all inflammasomes, and I shall concentrate on these here. The first point to note is that the IF (fed up of typing inflammasome already!) is an intracellular complex that promotes the "activation" of proteases and the maturation of cytokines. The immune system is littered with "latent" or "pro" forms of proteins, that require some form of modification, such as the release of an N-terminal peptide, before they can perform their inflammatory-related role. Think of trypsinogen and chymotrypsinogen, pro-enzyme forms of the serine proteases, trypsin and chymotrypsin respectively. The conversion of an inactive form to an active one by limited proteolysis or ligand binding is a common thread in biochemical regulation: the latter is referred to as "allostery" (discussed in an earlier post on RNA Polymerase).
 The image on the left, illustrates the protease cleavage events associated with the activation of Caspase 7, an enzyme (an executioner enzyme, no less) that is intimately associated with programmed cell death (apoptosis). Using cryo-electron microscopy (as an aside, you may have noticed how often this technique is appearing in the structural biology world: it seems to be an unstoppable force and is a major new focus at Sheffield) the authors derive a series of incredibly detailed molecular images. If you are looking for more information, the Wu lab home page contains links to publications that provide a comprehensive background to the molecules and their function. See here, for example.
The image on the left, illustrates the protease cleavage events associated with the activation of Caspase 7, an enzyme (an executioner enzyme, no less) that is intimately associated with programmed cell death (apoptosis). Using cryo-electron microscopy (as an aside, you may have noticed how often this technique is appearing in the structural biology world: it seems to be an unstoppable force and is a major new focus at Sheffield) the authors derive a series of incredibly detailed molecular images. If you are looking for more information, the Wu lab home page contains links to publications that provide a comprehensive background to the molecules and their function. See here, for example. Perhaps the most significant finding of this work (summarised schematically alongside related complexes on the left); notwithstanding the elegant microscopy, is the detail that has revealed how the IF is triggered by a single molecular recognition event from an invading bacterium (the pink blob in the image above), which in turn promotes the assembly of the intact and active IF. In addition, mutations in the NLRC4 protein (the pale blue wheel-shaped assembly attached to the pink cap, above) cause severe autoimmune inflammatory disease. The structure provides insight into how the IF fails to assemble properly in patients carrying these genetic lesions.The use of cryo-electron microscopy, drawing on X-ray crystallography has led to a molecular explanation for the triggering of the inflammatory response and moreover how mutations can throw a spanner in the works of macromolecular assembly, with such a devastating effect.
Perhaps the most significant finding of this work (summarised schematically alongside related complexes on the left); notwithstanding the elegant microscopy, is the detail that has revealed how the IF is triggered by a single molecular recognition event from an invading bacterium (the pink blob in the image above), which in turn promotes the assembly of the intact and active IF. In addition, mutations in the NLRC4 protein (the pale blue wheel-shaped assembly attached to the pink cap, above) cause severe autoimmune inflammatory disease. The structure provides insight into how the IF fails to assemble properly in patients carrying these genetic lesions.The use of cryo-electron microscopy, drawing on X-ray crystallography has led to a molecular explanation for the triggering of the inflammatory response and moreover how mutations can throw a spanner in the works of macromolecular assembly, with such a devastating effect.